Detoxification 101
Toxicology is one area of medicine about which clinical ecologists are not in conflict with their more conventional colleagues. We share the same issues, pursue the same phenomena and agree on therapeutic approaches. Toxicologists tend to veer in the direction of epidemiological effects, studying whole-groups, whereas the clinical ecologist takes it more patient by patient, but that?s the only real difference.
Toxicology has been around a surprisingly long time. Primitive peoples used natural poisons for hunting. Indeed the word toxicology comes from toxicos, the bow from which poison arrows were flung. The ancient Greeks and Romans made a special study of poisons, although more in connection with political assassinations than the pursuit of science. The Persian King Mithridates was so afraid of being poisoned that he took a regular cocktail of known poisons to accustom his body to their effect so that they would no longer work on him. From his name we get the word mithridate.
The prolific use of poisons for getting rid of “inconvenient” people led to a treatise by Maimmonides (1135-1204) entitled Poisons and their Antidotes. It summarized all knowledge of poisons at that time.
The Italian fifteenth-century Borgia family were infamous poisoners. Lucrezia?s name, in particular, achieved evil notoriety for her nefarious use of chemicals to “alter” history.
Toxicology finally adopted a more formal scientific footing, and today we are concerned almost entirely with environmental hazards and unintentional harm done to human beings. Around 4,000,000 man-made chemicals have been described in scientific literature since 1965. Something like 6,000 new chemicals are added to the list every week and at least 70,000 are currently in production. Only a fraction of this toxic load has been adequately tested for the long term effects on human health.
We meet chemicals in the air, our food, water supplies and by direct contact. Medical drugs add their share, and even the clothes we wear and the fabric of our homes are mostly artificially made, needing many complex chemical precursors. Some of these emit toxins long after being installed in the home. It is a fact of life in the modern world that indoor pollution can be just as bad, or worse, than the outdoor kind.
The cumulative effect of all these substances may create a total body burden that triggers chemical sensitivity in certain individuals. In the late 1970s Dr. E.C. Hamlyn coined the term ?human canary? to describe such people ? they are a warning to us all that we are going to be ill if we continue as we are, in much the same manner that canaries used to warn miners of impending gas danger. Unfortunately, no one seems to be heeding these canaries.
Most studies done on humans to date have been concerned predominantly with acute massive exposures suffered by workers in industrial settings, but clinical ecologists have been gathering case studies steadily to show that chronic exposure to levels commonly thought to be ?safe? are compromising people?s health and may turn out to be a more important hazard in the long term.
The effects of chemical exposure are dependent upon a number of factors, principally:
- the amount and biological activity of the compound
- length of the exposure time
- genetic factors
- biochemical individuality
- the total stress load
- age and sex
- previous exposures
- nutritional factors
The resulting problems can be complex, depending on the target organs involved. Misdiagnosis and missed diagnosis are the norm. Safety levels are misleading, since they are based on averages. Some individuals will react to far lower levels than would affect the majority.
The way the body disposes of unwanted and toxic compounds (xenobiotics) we call detoxification. In fact, the metabolic pathways discussed here, by which chemicals are inactivated and removed from the body, don?t always result in a less poisonous end-product. A better term, therefore, is biotransformation.
There are several pathways involved. The subject is a vast and burgeoning one; the information given here is necessarily selective.
Metabolism of toxic compunds
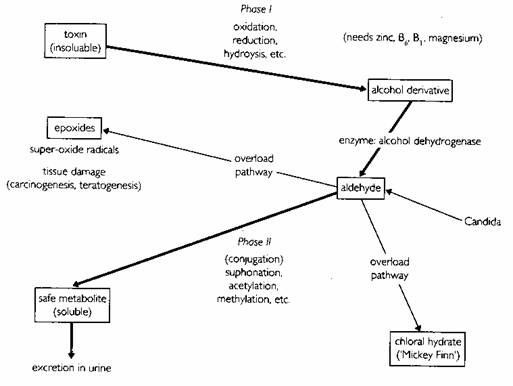
To get rid of a toxin effectively it is most important that the body turns it into something soluble in water. At that point the substance or its metabolites (breakdown products) can be removed via the kidneys, sweat, bile and other fluids. There are two principal routes by which the body does this. In Phase I metabolism the molecule is altered by enzymes in a variety of ways, each process assisted by a specific enzyme. These enzymes are found in the microsomes of most cells.
The most important of these enzyme pathways is the cytochrome P450 system, also called the multi-function oxidase system (MFO). Under its influence oxygen is added to the toxic molecule, converting a hydrogen atom in the molecule into a hydroxyl group (hydrogen and oxygen). The opposite effect, known as reduction, means that hydrogen is added. Both effects can knock out the toxicity of a molecule.
Two other methods to note are: adding a water molecule (2 hydrogen and 1 oxygen atoms), called hydration and knocking out halogen atoms, such as chlorine, called dehalogenation.
It is important to note that magnesium is essential for Phase I actions, as is a complex co-enzyme called nicotinamide adenine dinucleotide (NADH), a derivative of vitamin B3. Vitamin C and zinc are also said to help, and possibly other nutrients as well. This is why vitamin and mineral supplements can be so vital for allergics and poor metabolizers.
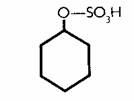
Phase II detoxification is carried out differently. Here extra groups are stuck on to the basic molecule. These change its character and render it harmless and more soluble. We call this process conjugation. An example is sulphation, the addition of a sulphate group (-SO3).
Phenol (carbolic acid), which looks like this: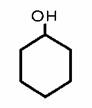
Is converted into phenyl sulphate, which looks like this:
The enzyme in this case is sulphonyl transferase. Phenol sulphonyl transterase may have great importance for food intolerance since it has become clear that a number of foods contain several phenolic compounds.
A study published in January 2004 found pollution, in particular, diesel exhaust, could significantly worsen symptoms in allergy sufferers. The study, which appeared in The Lancet, showed that when patients were exposed to diesel exhaust in addition to an allergen, histamine production increased by five-fold.
And these effects were magnified in people with a particular genetic makeup. People with a mutation to the gene responsible for making an enzyme known as glutathione S-transferase M1 (GSTM1) responded even more strongly to the combination of diesel exhaust and allergen, the researchers found.
As many as 50% of people are born with this mutation, says the study’s lead author, Dr. Frank Gilliland, a professor of preventive medicine at the Keck School of Medicine at the University of Southern California.
The Asthma Connection
Recent research has also implicated pollution in asthma development and exacerbation.
For example, a study published in January in the Journal of Epidemiology and Community Health found that children who spent their first three years near a busy highway were more likely to develop asthma than those who lived near quieter streets.
And a report published in November 2003 in the Journal of Allergy & Clinical Immunology showed that diesel exhaust could trigger an asthma-like episode in mice.
What does this mean for allergy and asthma sufferers?
You need to watch both the pollution levels as well as the pollen levels, experts say.
“This is a big issue for us in Los Angeles. We don’t have the greatest air quality,” author Riedl says. “I tell my patients to watch the news for the air quality report. And if the air quality is poor or even moderate, they need to be aware of that.”
And on those really bad days, it may make sense to minimize exposure to dirty air.
“For people with asthma or serious allergic problems, it’s best to stay inside if they can,” Riedl says. “And if you have to be outside, avoid strenuous exercise.”
If you do need to go outside, you might want to wear a mask that covers your nose and mouth, says Dr. Marjorie Slankard, an associate professor of medicine at the Columbia University College of Physicians and Surgeons in New York.
Side routes of detoxification
These optimal biotransformation pathways can be blocked for a number of reasons. Certain vitamin and mineral deficiencies could do it; magnesium, for example, has been mentioned. Overload can have the same effect. As the total quantity of xenobiotics increases, we can produce more of the relevant enzyme, up to a point (this is called enzyme induction). But eventually we pass the equalization point and the body can no longer cope.
When the basic system is no longer capable of keeping pace, ?alternative? toxic metabolites may be chosen which may be more stable and can?t easily be brought back into the enzyme pathways and broken down. Some of these ?alternatives? are capable of causing serious tissue and gene damage.
Mickey Finn
Less serious, perhaps, but troublesome nevertheless is the alternative pathway that yields chloral hydrate. At times of overload, this chemical can build up. It is one of the ingredients of the classic “Mickey Finn” . If this build-up occurs the patient will begin to feel very fatigued and “spacey”. These are symptoms that overload patients will recognize.
Candida, incidentally, is capable of producing alcohols and aldehydes, which well add to the overload of these detoxification pathways.
The diversity of routes for xenobiotics and the consequences that they may have for the organism can be represented diagrammatically, as shown in the next figure. ?
Fast ans slow metabolizers fast and slow metabolizers
There is a sulphoxidation reaction test the can detect the amount of sulphoxide in the urine following a loading dose of a suitable test substrate.
Those who don?t handle xenobiotic chemicals very well produce less metabolite output to the urine. We can call them ?slow metabolizers?. Preliminary results show that the white European population has less than 20 per cent slow metabolizers.
A genetically-determined ability to metabolize in this way could be an important factor in a person?s response to toxic environmental chemicals and could deterrmine whether he or she will ultimately contract cancer. It is now held likely that poor metabolizers are the people who suffer from chemical intolerance and are those most likely to become universal reactors or to develop ?twentieth-century allergies?.
The implication of al this is startling, to say the least. If it turns out to be correct it means that many diseases may have a basis in chemical overload. For example, the incidence of Parkinson?s disease is found to have a surprising and extraordinarily exact correlation with areas of high pesticide use and also affects a larger-than-expected percentage of slow metabolizers. Could it be an ecological illness?
What can you do?
First and foremost, you need to reduce your xenobiotic load. This is covered in the Chemical Clean-up section.
Secondly, get yourself onto an adequate supplement programme, to counter the effects of these toxic substances and to aid the body’s ability to detoxify. Sherry Rogers, whose brilliant book TIRED OR TOXIC I highly recommend, gives the following regime.
Nutrient Rationale
Multiple minerals (with iron, chromium, molybdenum, vanadium, selenium, copper, zinc, magnesium, potassium, calcium, iodine, cobalt and silicon: it’s unlikely you will get one with all of these) people get progressively fewer minerals from their diet.
Antioxidant (with A, C, E, l-cysteine or n-acetyl-cysteine, glutathione, selenium, B complex with extra B3 (or NADH), PABA, beta-carotene, alpha-lipoic acid) most people are in a free radical overload. Xenobiotic chemicals increase your free radical burden and deplete your anti-oxidants at the same time.
Multiple B (B1, B2, B3, B5, B6, B12, folic acid, PABA, inositol, choline, biotin This group are commonly low and consequences are serious.
Multiple vitamin/mineral preparations (beware: some have strange imbalances. Avoid ones with over 2 mgm copper a day, less than 5 mgm of manganese, less than 15 mgm of zinc, or a magnesium to calcium ratio of less than 3:4. Be sure to tally with multiminerals. To balance out the others.
Essential fatty acids, especially omega-3. Make sure the oil is in dark opaque, dated bottle, to protect from light oxidation Most people are hopelessly low on EFAs, and proportionately down on omega-3s.
There are of course many other good nutrients and support, including Chinese herbs, amino acids, digestive enzymes and homeopathic (homotoxicology) remedies.